|
11/21/2023 0 Comments Sigma bonds in co2Calculating Hybridisationsįirstly, let us find out the types of bonds in the molecule by making the chemical structure of the molecule. 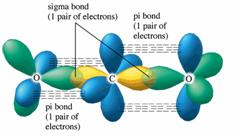
However, only 1 sp hybrid orbital will be used from these 3 sp hybrid orbitals to make a bond with carbon atoms. Moreover, the p orbital inside the atom of oxygen stays the same and is used primarily to create a pi-bond. The oxygen hybridised its orbital for creating 3 hybrid orbitals of sp2 in the CO2 molecule. A pi-bond is formed between the 2 leftover p electrons. 
These hybridised sp orbitals belonging to the carbon atoms extend beyond 2p orbitals that belong to the atoms of oxygen for creating two sigma bonds. Therefore, one electron from the 2s orbital shifts from the 2s level to 2p level, which leads to the creation of 2 hybrid orbitals. However, this is not sufficient for creating bonds involving the oxygen. 
The atom of the carbon comprises 2 double bonds, i.e., 2 effective pairs exist in it. Examples have been illustrated in Table 2.For determining carbon dioxide’s hybridisation, firstly let’s take the atom of carbon. E.g.: In cyclooctatetraene (C 8H 8), Y = 8, therefore A c = 24/2 = 12 number of single bonds. Where A c = number of single bonds and y is number of hydrogen atoms in aliphatic cyclic olefin. The total number of single bonds in aliphatic cyclic olefin can be calculated by using the formula Eg: In cyclooctatetraene (C 8H 8), X = Y = 8, therefore S c = 8+8 = 16 number of σ bonds. Where, X = number of carbon atoms Y = number of hydrogen atoms and S c = number of sigma bonds (σ-bonds) in cyclic olefinic system. The formula to calculate the number of σ bonds for an aliphatic cyclic olefin is In the first case, we have to count the number of carbon atoms (X) and the number of hydrogen atoms (Y) in the given unsaturated cyclic olefinic hydrocarbons. E.g.: In cyclooctatetraene (C 8H 8), X = Y = 8, therefore P c = 16-8/2 = 4 number of π bonds or double bonds. Where, X = number of carbon atoms Y = number of hydrogen atoms and P c = number of π bonds or double bonds in the cyclic olefinic system. The formula to calculate the number of π bonds or double bonds for an aliphatic cyclic olefin is Table 1: Calculation of π-bonds, σ-bonds, single and double bonds in open chain olefinic hydrocarbonsĬalculation of π-bonds and double bonds (Pc): Examples have been illustrated in Table 1. Where A = number of single bonds and Y is number of hydrogen atoms. The total number of single bond for an aliphatic straight chain olefin is Where, X = number of carbon atoms Y = number of hydrogen atoms and S = number of sigma bonds (σ-bonds). The formula to calculate the number of σ bonds for an aliphatic straight chain olefin is In this case, first we have to count the number of carbon atoms (X) and the number of hydrogen atoms (Y) in the given unsaturated hydrocarbon containing double bonds. 
Where, X = number of carbon atoms Y = number of hydrogen atoms and P = number of π bonds/double bonds. The formula to calculate the number of π bonds or double bonds for an aliphatic straight chain olefin is In the first case, we have to count the number of carbon atoms (X) and the number of hydrogen atoms (Y) in a given unsaturated hydrocarbon containing double bonds. \)Ĭalculation of π-bonds and double bonds (P):
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
